From May to September of 2014, I was part of a five-person team at Cardica in the engineering-build phase of an emerging surgical stapler.
Machine Design Engineering
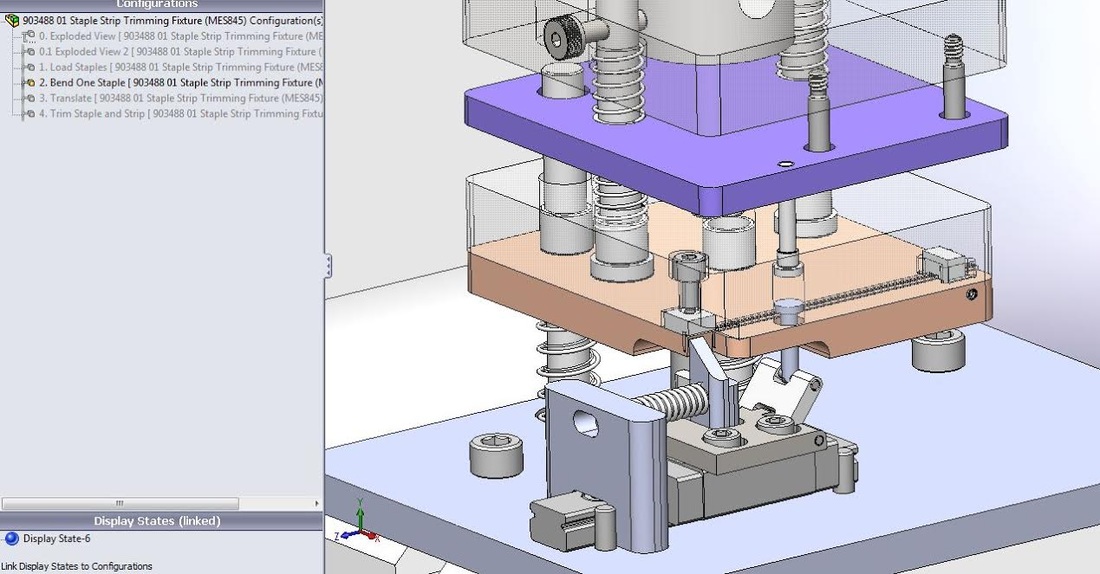
As our team moved into production, we designed fixtures for the assembly line. I designed a production fixture to rotate, form, translate, and cut staples before they were placed in the staple cartridge. I modeled the fixture with Solidworks, using four configurations to demonstrate a spring-based timing mechanism within an arbor press. I also emphasized design for manufacturability by designing with machinists. I personally milled 25% of the parts, and the rest of the fixture was outsourced to local machine shops.
Testing
One of the major goals of the engineering-build phase was to increase the strength of our components. I worked on a variety of testing projects in order to help our team pursue this goal. For example, I conducted engineering notebook tests using an Instron machine to increase stapler device clamp force by analyzing cable creep, injection molded component strength, and assembly fixture accuracy. I also increased the maximum angle of stapler articulation by perfecting the balance of friction and clearance between the plastic housing, steel gears, and steel bands by laser welding shims and improving assembly constraints.
Machine Design Engineering
As our team moved into production, we designed fixtures for the assembly line. I designed a production fixture to rotate, form, translate, and cut staples before they were placed in the staple cartridge. I modeled the fixture with Solidworks, using four configurations to demonstrate a spring-based timing mechanism within an arbor press. I also emphasized design for manufacturability by designing with machinists. I personally milled 25% of the parts, and the rest of the fixture was outsourced to local machine shops.
Testing
One of the major goals of the engineering-build phase was to increase the strength of our components. I worked on a variety of testing projects in order to help our team pursue this goal. For example, I conducted engineering notebook tests using an Instron machine to increase stapler device clamp force by analyzing cable creep, injection molded component strength, and assembly fixture accuracy. I also increased the maximum angle of stapler articulation by perfecting the balance of friction and clearance between the plastic housing, steel gears, and steel bands by laser welding shims and improving assembly constraints.
Material Strength Analysis
We also needed to choose new materials for various components. I performed FEA (AutoDesk) analysis of stress and strain of staple cartridge’s assembly containing multiple stainless steel and plastic over-molded components. I also worked with a variety of stainless steel grades, choosing for cost, corrosion resistance, and strength, including considering the ability to heat treat.
Medical Device Procedures
I worked not only with the engineers in the R&D group, but in the machine shop, on the assembly line, and with the inspection department. I learned a lot from the wide variety of problem solving approaches. I iteratively assembled prototype devices with technicians, following clean room procedures and manufacturing instructions. I am familiar with manual and automated optical inspection. In particular, we used an OPG Smartscope affectionately known as the "Flash." I complied with quality system documentation as we worked towards 510(k) clearance. This included creating and managing the finished goods master list, manufacturing instructions (MI), bill of materials (BOM), and lot history records (LHR).
We also needed to choose new materials for various components. I performed FEA (AutoDesk) analysis of stress and strain of staple cartridge’s assembly containing multiple stainless steel and plastic over-molded components. I also worked with a variety of stainless steel grades, choosing for cost, corrosion resistance, and strength, including considering the ability to heat treat.
Medical Device Procedures
I worked not only with the engineers in the R&D group, but in the machine shop, on the assembly line, and with the inspection department. I learned a lot from the wide variety of problem solving approaches. I iteratively assembled prototype devices with technicians, following clean room procedures and manufacturing instructions. I am familiar with manual and automated optical inspection. In particular, we used an OPG Smartscope affectionately known as the "Flash." I complied with quality system documentation as we worked towards 510(k) clearance. This included creating and managing the finished goods master list, manufacturing instructions (MI), bill of materials (BOM), and lot history records (LHR).

